

Get access to all handy features included in the IVIS website
- Get unlimited access to books, proceedings and journals.
- Get access to a global catalogue of meetings, on-site and online courses, webinars and educational videos.
- Bookmark your favorite articles in My Library for future reading.
- Save future meetings and courses in My Calendar and My e-Learning.
- Ask authors questions and read what others have to say.
Peritoneum and Abdominal Wall
Get access to all handy features included in the IVIS website
- Get unlimited access to books, proceedings and journals.
- Get access to a global catalogue of meetings, on-site and online courses, webinars and educational videos.
- Bookmark your favorite articles in My Library for future reading.
- Save future meetings and courses in My Calendar and My e-Learning.
- Ask authors questions and read what others have to say.
Read
Closure of Abdominal Incisions
Eberhard Rosin
The most common surgical procedure in small animal practice is incision and closure of the abdominal cavity. Although use of simple interrupted sutures to appose the peritoneum and all fascial layers is the traditional method to close an abdominal incision, a simple continuous suture pattern for a single-layer closure of the rectus fascia, without concern for the peritoneum, is a faster and safe alternative. In paralumbar grid incisions, a layered simple continuous closure is used if the patient’s musculature is well developed.
Surgical Anatomy
The external leaf of the rectus sheath is composed of the aponeurosis of the external abdominal oblique muscle, most of the aponeurosis of the internal abdominal oblique muscle, and, near the pubis, a portion of the aponeurosis of the transversus abdominis muscle. The internal leaf of the rectus sheath consists of a portion of the aponeurosis of the internal abdominal oblique muscle, the aponeurosis of the transversus abdominis muscle, and the transversalis fascia. In the caudal third of the abdominal wall the internal leaf disappears. The aponeurosis of the internal abdominal oblique muscle joins the external leaf, and the rectus abdominis muscle is covered only by a thin continuation of the transversalis fascia and peritoneum (Figure 23-1).1

Figure 23-1. Cross sections showing the anatomy of the sheath of the rectus abdominis muscle in the cranial and caudal portions of the abdominal wall.
Healing of the Peritoneum
The peritoneum is a layer of flat cells, cemented edge to edge at their intercellular margins. This mesothelial layer is supported by an underlying layer of areolar tissue that blends with the connective tissue of the transversalis fascia. Within the peritoneal cavity, a small amount of serous fluid contains freely floating cells including macrophages, desquamated mesothelial cells, and small lymphocytes.2
During the first 2 days after wounding, the peritoneal defect is red, with a glistening, slightly irregular surface. During the next 5 or 6 days, the color fades gradually, the surface becomes smoother, and the defect develops a homogeneous, transparent gray sheen. Abdominal tissues slide readily over the defect. Gradually, this gray sheen becomes more opaque until, after 2 or 3 weeks, the area usually is indistinguishable from normal peritoneum. Milky streaks beneath the wound area, apparently resulting from scar formation, may remain. These changes occur simultaneously throughout the entire defect. Large wounds heal as rapidly as small wounds.2-4
Microscopically, defects in the peritoneum are covered rapidly by macrophages, which are present in large number in the peritoneal fluid bathing the wound surface. The wound also is invaded by monocytes and histiocytes from blood and underlying exposed tissues. Cells from peritoneal fluid, blood, or underlying tissues differentiate to form fibroblasts, and the superficially located cells undergo metaplasia, gradually forming mesothelial cells. At the same time, intact mesothelial cells at the perimeter of the wound help in the repair by proliferation and migration. Small defects in the peritoneum are healed by proliferation of adjacent mesothelial cells, whereas large defects are covered by undifferentiated cells that then become mesothelial cells.2-4
Peritoneal defects that are sutured have a higher incidence of adhesions than defects that are left open to heal. The stimulus for adhesion formation is not the peritoneal defect itself, but rather the ischemic tissue that results when edges of the defect are brought together by sutures.
No evidence, experimental or clinical, supports the contention that closure of the peritoneum is necessary for wound strength, to minimize postoperative dehiscence or hernia formation, or to minimize the development of adhesions. On the contrary, experimental and clinical studies in dogs, horses, and human patients indicate that suturing the peritoneum should be avoided to minimize the incidence of postoperative intra-abdominal adhesions.3-7
Closure Alternatives
Closure of the Internal and External Leaves of the Rectus Sheath Versus Closure of the External Leaf Only
Closure of the paramedian abdominal incision by apposition of the internal and external leaves of the rectus sheath is traditional and has proved successful for years of clinical experience. However, closure of the internal leaf takes time and requires that the abdominal wall be manipulated to expose the internal leaf, which frequently retracts after incision. Studies have refuted the admonition that closing only the external leaf of the rectus sheath provides insufficient strength to the incision. In a biomechanical study of healing abdominal incisions in the dog, the strength of incisions closed by suturing the internal and external leaves of the rectus sheath and the strength of incisions closed by suturing the external leaf only were similar.8
Simple Interrupted Versus Simple Continuous Suture Pattern
The traditional method to close an abdominal incision is simple interrupted sutures. The same incision can be closed more quickly using a simple continuous pattern, with no difference in wound healing. In a randomized prospective trial of 3135 human patients comparing continuous and interrupted abdominal midline incision closure, no difference was found in the incidence of wound dehiscence.9 In clinical use of simple continuous closure of abdominal incisions in over 5000 dogs and cats, the incidence of dehiscence is negligible.
Closure Techniques
Midline Incision
With an incision through the linea alba in the cranial two-thirds of the abdominal hall, fibers of the rectus abdominis muscle are not exposed, and the linea alba, including the peritoneum, can be apposed accurately by full-thickness sutures. An adequate portion of fascia must be included with each suture, and the falciform ligament must not be interspersed between the edges of the linea alba (Figure 23-2). Although the traditional suture pattern is simple interrupted, a simple continuous pattern is a safe and faster alternative.
In the caudal third of the abdominal wall, the width of the linea alba decreases. An incision here frequently exposes the rectus abdominis muscle. Because fibers of the rectus abdominis muscle have little holding power, sutures are not full thickness. Instead, sutures are placed to include an adequate portion of the external leaf of the rectus sheath on each side of the incision and to appose this fascia accurately without interspersion of rectus abdominis muscle (Figure 23-3). The transversalis fascia and the peritoneum are not included in the sutures. Sutures traditionally are simple interrupted, but a simple continuous pattern is a satisfactory alternative.

Figure 23-2. Linea alba incision in the cranial two-thirds of the abdominal wall closed by a full-thickness suture placed carefully to avoid the falciform ligament.
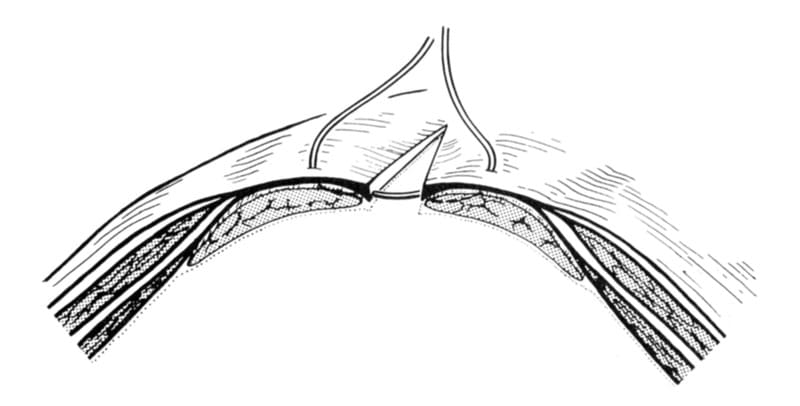
Figure 23-3. Midline incision in the caudal third of the abdominal wall closed by a suture placed to appose the external leaf of the rectus sheath accurately.
Paramedian incision
If the incision is paramedian in the cranial two-thirds of the abdominal wall, the linea alba will be on one side and the external and internal leaves of the rectus sheath and rectus abdominis muscle will be exposed on the other side, or on both sides of the incision, the internal and external leaves of the rectus sheath and rectus abdominis muscle will be exposed. The external leaf of rectus sheath is closed with a simple interrupted or continuous pattern. The internal leaf of the rectus sheath and the peritoneum are left unsutured (Figure 23-4).

Figure 23-4. Single-layer closure of only the external leaf of the rectus sheath. Care is taken to avoid interspersing rectus muscle between the edges of the rectus sheath.
A paramedian incision in the caudal third of the abdominal cavity is closed by suturing the rectus fascia in a simple interrupted or continuous pattern. The transversalis fascia and peritoneum have little strength and are not sutured.
Simple Continuous Suture Technique
Acceptable suture materials include polyglycolic acid, polyglactin 910, polydioxanone, polyglyconate, polypropylene, and nylon. Surgical gut, stainless wire, and multifilament nonabsorbable suture materials should not be used. Suture size is based on patient size: 3-0 suture material should be used for cats and small breed dogs, 2-0 for medium-sized dogs, 0 for large dogs, and 1 for giant breed dogs.
For a simple continuous suture pattern used in long incisions, more than one strand of suture material is used. Sutures are placed through the linea alba or through the external leaf of the rectus sheath, depending on patient size, and, include a 3- to 10-mm width of fascia on each side of the incision. Sutures are placed 5 to 10 mm apart, depending on the size of the animal. Care is taken to ensure edge-to-edge apposition of the fascia without interspersion of muscle. The internal leaf of the rectus sheath and the peritoneum are not included in the suture pattern.
All knots are placed with care. The first throw is tied with appositional tension only to ensure that tissue is not strangulated. Five additional square, flat throws are placed.10 After each throw is made, the ends of the suture are pulled tight to make the knot secure. The ends of the suture are cut 4 mm from the knot. As the continuous suture is placed, the rectus fascia must be loosely approximated, not apposed with tension. Wound strength is adversely affected if fascia is closed tightly.11
Subcutaneous tissues are closed with the same suture materials, usually a smaller size, placed in simple continuous pattern. Care is taken to avoid cutting the rectus fascia suture during closure of the subcutaneous tissue. Skin is closed with 3-0 nonabsorbable suture placed in a simple interrupted or cruciate pattern, or skin staples are used.
References
- Evans HE, Christensen GC. Miller’s anatomy of the dog. 2nd ed. Philadelphia: WB Saunders, 1979.
- Ellis H, Ashby EC, Mott TJ. Studies in peritoneal healing: a review. J Abdom Surg 1969,11:110.
- Hubbard TB, et al. The pathology of peritoneal repair: its relation to the formation of adhesions. Ann Surg 1967; 165: 908.
- Ellis H. The cause and prevention of postoperative intraperitoneal adhesions. Surg Gynecol Obstet 1971;133:497.
- Karipineni RC, Wilk PJ, Danese CA. The role of the peritoneum in the healing of abdominal incisions. Surg Gynecol Obstet 1976:142:729.
- Swanwick RA, Milne FJ. The non-suturing of parietal peritoneum in abdominal surgery of the horse. Vet Rec 1973:93:328.
- Ellis H, Heddle R. Does the peritoneum need to be closed at laparotomy? Br J Surg 1977;64:733.
- Rosin E, Richardson S. Effect of fascial closure technique on strength of healing abdominal incisions in the dog: a biomechanical study. Vet Surg 1987; 16:269.
- Fagniez P, Hay JM, Lacaine F, et al. Abdominal midline incision closure: a multicentric randomized prospective trial of 3,135 patients, comparing continuous vs interrupted polyglycolic acid sutures. Arch Surg 1985:120:1351.
- Rosin E, Robinson GM. Knot security of suture materials. Vet Surg 1989:18:269.
- Stone KI, vonFraunhofer JA, Masterson BJ. The biochemical effects of tight suture closure upon fascia. Surg Gynecol Obstet 1986,163:448.
Closed Peritoneal Drainage
Giselle Hosgood
Indications
The use of drains to remove fluid, pus, or contaminated material from the closed peritoneal cavity is hereby referred to as closed peritoneal drainage. The use of drains in the peritoneal cavity is primarily indicated in the management of peritonitis although there is some evidence to show that appropriate surgical and medical management without drainage is equally effective.1-4 Other indications for peritoneal drainage include diagnostic peritoneal lavage, peritoneal dialysis and administration of peritoneal chemotherapy.5 Placement of drains after routine abdominal procedures is discouraged, and the use of drains should not replace meticulous surgical technique. The use of drains can be associated with multiple complications, and peritoneal drainage is not a reliable indicator of wound or body cavity events; the absence of drainage does not always imply the absence of fluid and fluid production can be induced by the presence of the drain alone.1
Closed-collection systems
Connecting a drain to a collection system has several advantages, and is strongly recommended. A collection system, whether by passive or active drainage, reduces the risk of ascending infection through the lumen of the drain. Ascending infection is one of the most common complications of peritoneal drainage, a function of both bacterial load migrating up the drain and decreased local tissue resistance because of the presence of the drain.6 Although bacteria can also migrate along the outside of the drain,7 a closed system greatly reduces the bacterial load. Protection of the drain by a sterile bandage can reduce bacterial migration along the outside of the wound. Collection systems also eliminate the chance of saturating the bandage covering an open drain. A wet bandage over a freely draining passive drain provides an additional source of contamination by bacterial strike-through from the environment. Collection systems also allow accurate assessment of fluid character and volume.
Passive Drainage
The abdomen can be compared with a fluid-filled, flexible container with two separate pressure zones. Positive atmospheric pressure exists within the gastrointestinal tract, whereas the peritoneal cavity has an extraluminal subatmospheric pressure between -5 and -8 cm water; the pressure is most negative in the cranial abdomen near the diaphragm.8 Passive drainage of fluid from the peritoneal cavity requires an air vent to break the vacuum and to create a positive atmospheric pressure within the peritoneal cavity.8 Positive pressure occurs after celiotomy or after a stab incision into the abdomen for drain placement. Passive peritoneal drainage relies on a pressure differential between the peritoneal cavity and the environment and functions primarily by overflow. Passive drainage is gravity dependent, and the drain provides a tract of least resistance along which excess fluid flows.2,8 An inexpensive and easily accessible passive collection system is trough a sterile intravenous administration set into a sterile bag. Sterile urine collection systems can also be used. Although passive drains are gravity dependent,1 the resting level of the drain and the collection bag proportionally affect the gravitational force and the rate of drainage. Increasing this distance excessively may promote obstruction of the drain by omentum or intestinal loops; for instance, having a dog in a high cage with the collection bag on the floor.
Active Drainage
Active drainage requires an external vacuum to create negative pressure within the peritoneal cavity. This allows drainage to occur independent of gravity. The vacuum is achieved by connection of the drain to a compressible container or a constant, low-pressure, suction device (Figure 23-5).1,2 Some commercial collection systems have one-way valves to prevent fluid reflux from the collection system into the peritoneal cavity. Suction should be applied to the drain before complete abdominal closure to prevent occlusion of the drain by intraluminal blood clot formation or tissue debris. The optimal level of suction is unclear. Low-level suction is effective, but higher levels are not always harmful and may remove more fluid. Suction levels between -9 mm Hg (-12 cm water)9 and -15 mm Hg (-20 cm water)10 are typically used in wound drainage in people, although higher levels of suction, -80 mm Hg (-112 cm water)11 and -150 mm Hg (-200 cm water),12 have been used successfully. Closed active drains are effective in removing large volumes of fluid; however, as the volume of intraperitoneal fluid decreases, active drainage causes suction of tissue, viscera, omentum, or abdominal wall toward the drain, resulting in occlusion.1,13 Tissue debris may also occlude the lumen. High-level suction may promote obstruction.
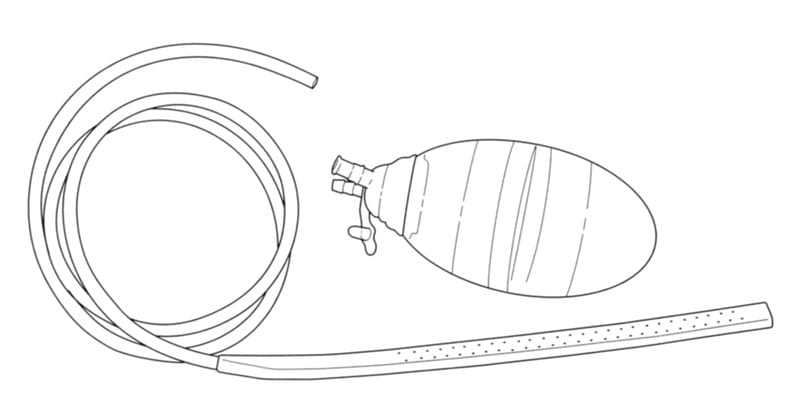
Figure 23-5. A silicone wound drain and compressible collection canister that can be connected together and used for active peritoneal drainage. Note the one-way valve on the canister. Alternately, the drain can be connected to a passive collection system.
Drains Suitable for Peritoneal Drainage
The most suitable drain for use in the peritoneal cavity is a simple tube drain with multiple fenestrations. Variations include channel drains and multilumen drains with channels (Figure 23-6).These tube drains can be connected to an active or passive collection system. Alternate techniques of open peritoneal drainage for the management of pancreatic masses5 and peritonitis3 are also reported.

Figure 23-6. Cross-sectional images of multifenestrated silicone wound drain A., Channel drain B. and multilumen drain with channels C.
Simple Tube Drain
The simple tube drain is a single lumen drain that acts primarily by gravity-dependent intraluminal flow. Because most of the drainage is intraluminal, fenestration improves drainage efficiency.14 However, fenestration reduces the tensile strength of the drain and may predispose to tearing on removal.15 Channel drains function by extraluminal capillary flow along the channels of the drain. The channelling increases the surface area of the drain, thereby increasing the efficiency. Multilumen drains with channels function by both intra and extraluminal flow whereby channels empty into the lumens via multifenestrations along the length of the drain. Presumably, channelling reduces tissue occlusion against fenestration of the simple tube drains. Pre-fenestrated commercial drains are preferred but fenestrations can be made by hand in a solid drain. Fenestrations should be oval and less than one-third the diameter of the drain, to prevent kinking and tearing.6 Fenestration using oval-tipped bone rongeurs rather than scissors may give a more precise, controlled cut with easy and safe removal of the fragment in the instrument’s jaws.16 Channel drains can only be purchased. Silicone drains are preferred over plastic (polyvinyl chloride) or rubber drains because silicone is relatively inert (red rubber tubes are the most irritating) and the silicone is soft and pliable. However, silicone drains have less tensile strength than polyvinyl chloride drains, and care is required on removal to prevent tearing.15 Silicone wound drains are readily available (Cardinal Health Fluid Management Products, 1500 Waukegan Road McGaw Park, IL 60085)
Peritoneal Dialysis Catheter
A modification of the simple tube drain is the multi-fenestrated peritoneal dialysis catheter (Quinton Tenckhoff Peritoneal Catheter, Kendal Healthcare, Covidien, Mansfield, Massachusetts; Figure 23-7).

Figure 23-7. A commercial, multifenestrated, peritoneal dialysis catheter. Note the Dacron cuff toward the end of the drain and the radiopaque ridge on top of the drain.
Most peritoneal dialysis catheters are now made of silicone. Some peritoneal dialysis catheters have one or two Dacron cuffs around the tubing that is sutured to the body wall and subcutaneous tissue to stimulate a fibroblastic reaction and to prevent subcutaneous fluid leakage. These catheters are also used for intraperitoneal chemotherapy. Some peritoneal dialysis catheters come with an introducer or trochar which allows for easy percutaneous insertion into the peritoneal cavity. These types of peritoneal catheter are particularly useful for diagnostic peritoneal lavage or intraperitoneal administration of drugs because they can be inserted through a small abdominal incision using local anesthesia in a sedated animal.5
Drain Placement
Efficient peritoneal drainage is difficult because of the convoluted nature of the peritoneal cavity, the sometimes intense fibrinous and fibrous reactions encountered during peritonitis, and the ability of the omentum to isolate the drain from the peritoneal cavity. Normal forces associated with movement of the diaphragm, abdominal wall, and intestine affect the intraperitoneal circulation of fluid and cause fluid to pool beneath the diaphragm16 and in the peritoneal reflections near the distal colon. In addition to normal forces, fluid can become isolated by peritoneal adhesions.9 Drain placement is extremely important to maximize drainage. Drains are usually placed during celiotomy. Alternately, in acute situations such as diagnostic peritoneal lavage, emergency drainage of fluid from the abdomen (uroabdomen), or for establishment of peritoneal dialysis, a stab incision in the skin of the ventral midline is made, and the drain is “punched” through the body wall. Commercial, simple, peritoneal dialysis catheters and some tube drains come with a stylet-trocar for this purpose. Insertion of these drains in an emergency can often be performed under local anesthesia and sedation.
To drain the peritoneal cavity effectively, two tubular drains may be required, one directed cranially along the ventral abdominal wall toward the diaphragm and the other caudally along the ventral abdominal wall to the peritoneal reflections near the distal colon.9 Alternately, commercial drains are available that “split”, allowing the two sections of the drain to be placed in different directions with the advantage of only having a single collection tube and exit point. The drains should exit close to the midline, in a dependent position, usually between the umbilicus and the xiphoid. The exit incision through the abdominal wall and skin should be only as large as the diameter of the drain.
A small exit incision reduces subcutaneous fluid leakage and subsequent cellulitis and possible incisional herniation. The exit incision should not be so small as to obstruct the drain. The epigastric vessels, which run through the middle of the mammary chains, should be avoided. If the exit incision is small, it can be made between the epigastric vessel and the midline (incision) without weakening the midline celiotomy incision. Exiting lateral to the epigastric vessels moves away from a dependent exit site and may promote subcutaneous fluid leakage and cellulitis. If celiotomy is performed, tacking the omentum to the stomach may help to prevent it from enveloping the drains. The omentum can be excised if it is obviously compromised or heavily contaminated. Excising the falciform fat may help to reduce tissue obstruction of the drains.
The exit tubing can be connected to a collection system, to function by passive drainage or low-pressure suction (40 to 60 mm Hg). All drains should be sutured to the skin using a secure suture such as the Chinese finger trap suture.18 Suction is applied as soon as the drain is placed, to prevent intraoperative obstruction of the drain with blood clots or tissue debris. All drain exit points should be covered with a sterile bandage on completion of the surgical procedure. If for some reason a closed system is not used, multilayered, thick, absorbent bandage material is indicated to cover the drain, noting that the contact layer must be sterile. Sterile cloth towels and sterile disposable diapers (sterilized with ethylene oxide) make useful absorbent bandage layers.
Postoperative Management
The bandage should be changed as often as required to prevent complete soaking by exudate and possible strikethrough of bacteria from the environment. The frequency of bandage changes is considerably reduced for drains using a collection system, but some leakage can occur through the exit site around the drain. Contamination of the bandage from the environment (urine, feces) can also occur. Use of an indwelling urinary catheter, particularly in male dogs, may help to prevent urine contamination. This is especially useful if the animal is recumbent. Bandaging also helps to prevent self-mutilation of the drainage area and premature removal or damage to the drain by the animal. The volume and nature of the fluid should be monitored closely, at least three to four times a day or more if profuse. The collection system should be changed using sterile technique when it is full or the vacuum has been lost. The vacuum may be lost before the collection system is completely full. Without vacuum and fluid flow, the risk of obstruction of the drain by tissue debris or fibrin and blood clots is increased. In addition, fluid that remains in the collection system for a prolonged period may promote bacterial growth. The drain is removed once the volume of fluid becomes significantly reduced and the fluid becomes serosanguineous. Drainage beyond 2 to 3 days is rarely necessary and persistent fluid production may indicate non-resolving peritonitis or other problems with response to management. The presence of a drain incites an inflammatory reaction and some fluid production (2 to 4 mls/kg/day), hence drainage usually does not cease completely. If drainage ceases suddenly, it may represent drain obstruction rather than resolution of the disease. Fluid may continue to drain for 1 to 2 days after drain removal, and a bandage should remain in place over the exit point to collect drainage and to prevent contamination of the exit site until it is sealed.
Complications
The most common complications of abdominal drains are obstruction and ascending infection. Nosocomial bacterial contamination of the drain and drainage site is a common complication of any drain placement. Retraction of the drain may occur once the animal begins to move and stand. This tends to occur with drains that are not connected to a collection system and that are cut short at the exit site. Suturing the drain securely, at several sites, is also important. Exit site and drain tract cellulitis is not uncommon. This complication is not serious and usually resolves once the drain is removed.
Subcutaneous fluid leakage is more common when peritoneal lavage is used and is particularly noted in the first few hours after surgery. Subcutaneous fluid leakage and cellulitis can be reduced by using a short subcutaneous tunnel between the skin and the abdominal wall exit site for the drain and by having the exit site in a dependent position. The Dacron cuffs on the peritoneal dialysis catheter and disc catheter also help to reduce this complication. Applying a water-repellent ointment to the skin around the exit site (petroleum jelly) may help to prevent skin irritation from drainage fluid. Hypoproteinemia and hypoalbuminemia are significant complications of peritonitis and drainage, but are not complications of drainage per se. Close monitoring of plasma protein concentrations in animals with peritonitis is imperative, and intravenous plasma or colloid infusion may be required.
Negative PressureWoundTherapy – Modified Closed Drainage
A modified application of suction drainage for septic peritonitis is the use of negative pressure wound therapy.19,20 The septic abdomen is essentially treated as an open wound with removal of exudate through the application of a sealed, absorbant dressing over the open abdomen which is connected to a commercial, portable suction apparatus. The suction apparatus provides continuous subatmospheric pressure at -80 mmHg to -125mmHg. While the abdomen is open, the drainage system is a closed system, isolated from the environment. Once infection is controlled, delayed abdominal closure is then performed.
References
- Donner GS, Ellison GW. The use and misuse of abdominal drains in small animal surgery. Compend Contin Educ Pract Vet 1986;8:705-715.
- Hosgood G. Drainage of the peritoneal cavity. Compend Contin Educ Pract Vet 1993;15:1605-1617.
- Staatz AJ, Monnet E, Seim HB 3rd. Open peritoneal drainage versus primary closure for the treatment of septic peritonitis in dogs and cats: 42 cases (1993-1999). Vet Surg 2002;31(2):174-80.
- Lanz OI, Ellison GW, Bellah JR, Weichman G, VanGilder J. Surgical treatment of septic peritonitis without abdominal drainage in 28 dogs. J Am Anim Hosp Assoc 2001;37(1):87-92.
- Hunt CA. Diagnostic peritoneal paracentesis and lavage. Compend Contin Ed Pract Vet 1980;11:449-453.
- Hampel NL, Johnson RG. Principles of surgical drains and drainage. J Am Anim Hosp Assoc 1985;21:21-28.
- Raves JJ, Slitkin M, Diamond DL. A bacteriologic study comparing closed suction and simple conduit drainage. Am J Surg 1984;148:618-620.
- Gold E. The physics of the abdominal cavity and the problem of peritoneal drainage. Am J Surg 1956;91:415-416.
- Tenta LT, Maddalozzo, Friedman CD, et al. Suction drainage of wounds of the head and neck. Surg Gynecol Obstet 1989;169:558.
- Kern KA. Technique for high volume drainage beneath large tissue flaps. Surg Gynecol Obstet 1990;170:70.
- Garcia-Rinaldi R, Defore WW, Green ZD, et al. Improving the efficiency of wound drainage catheters. Am J Surg 1975;130: 372-373.
- Moss JP. Historical and current perspectives on surgical drainage. Surg Gynecol Obstet 1981;152:517-527.
- Formeister JF, Elias EG. Safe intra-abdominal and efficient wound drainage. Surg Gynecol Obstet 1976;142;415-416.
- Hanna EA. Efficiency of peritoneal drainage. Surg Gynecol Obstet 1970,131:983-985.
- Paton RW, Powell ES. Which drain? A comparison of the tensile strengths of vacuum drainage tubes. J R Coll Surg Edinb 1988;33:127-129.
- Arnstein PM. Custom tube drains. Lancet 1988;1:215.
- Hosgood G, Salisbury SK, Cantwell HD, et al. Intraperitoneal circulation and drainage in the dog. Vet Surg 1989;18:261-268.
- Smeak DD. The Chinese finger trap suture technique for fastening tubes and catheters. J Am Anim Hosp Assoc 1990;26:215-218.
- Cioffi KM, Schmiedt CW, Cornell KK, Radlinsky MG. Retrospective evaluation of vacuum-assisted peritoneal drainage for the treatment of septic peritonitis in dogs and cats: 8 cases (2003-2010). J Vet Emerg Crit Care 2012: 22: 601-609.
- Buote NJ, Havig ME. The use of vacuum-assisted closure in the management of septic peritonitis in six dogs. J Am Anim Hosp Assoc 2012;48:164-171.
Omentum as a Surgical Tool
Giselle Hosgood
Introduction
The omentum is a mesothelial membrane with a unique vascular and lymphatic network that supports its use in various abdominal and extraabdominal surgical procedures. In any surgical procedure where there is a need for increased vasculature, lymphatics, or tissue bulk, the omentum may prove useful.1
Anatomy and Physiology2
The peritoneum is a mesothelial membrane that lines the abdominal cavity and covers the the abdominal organs. The peritoneal folds which leave the greater and lesser curvatures of the stomach are known as the greater and lesser omentum, respectively. The lesser omentum is derived from the ventral mesogastrium and extends between the lesser curvature of the stomach and the initial part of the duodenum to the liver hilus. The greater omentum is derived from the dorsal mesogastrium and comprises the large bursal portion and the smaller splenic and veil portions. Both omenta are composed of a double peritoneal leaf that is transparent, except for the lacey appearance of fat around the blood vessels that run through it.
The bursal portion of the greater omentum is of most clinical and surgical significance and is hereafter referred to as the omentum. The bursal portion is attached cranioventrally to the greater curvature of the stomach and extends caudally to the urinary bladder. The omentum reflects on itself forming a double layer (visceral and parietal layer) that covers the intestines (Figure 23-8). The potential cavity between the layers is the omental bursa (lesser peritoneal cavity). The only natural opening of the omental bursa is the epiploic foramen.
Important anatomical features of the omentum in the dog, which differ from that of people, may affect the surgical extension procedures that are sometimes performed depending upon its surgical use. In the dog, the spleen is attached to the parietal layer of the omentum. There is no colonic attachment of the omentum in the dog.1 The primary omental blood supply comes from right and left border vessels that arise from the right gastroepiploic and splenic arteries, respectively.3 Approximately nine smaller vessels originate from the gastroepiploic arcade along the greater curvature of the stomach.1 The gastroepiploic arcade does not require mobilization in the dog.1 The omentum is one of the major fat repositories in obese animals. Lymphatic drainage occurs by blind, bulbous capillaries present in the milk spots on the surface of the omentum. The mesothelial membrane is discontinuous over the milk spots, allowing material access to the lymphatics.4 Lymphatics follow the vascular paths and anastomose with lymphatics of the stomach and spleen which drain by regional and celiac lymph nodes into the thoracic duct.4 Drainage into the lacunae on the visceral surface of the diaphragm also occurs.2 Milk spots are collections of cells of lymphoid and myeloid origin, mainly T and B lymphoctes, monocytes and macrophages. These sites may provide cells that have roles in inflammation, angiogenesis and immune responses.

Figure 23-8. Peritoneal reflections of the dog in sagittal section. The dog is on its back, head to the right. (From Evans HE. The abdomen. Miller’s Anatomy of the Dog. Philadelphia: W.B. Saunders Co; 1993:434).
Surgical Techniques
Mobilization of the Omentum
For use in local abdominal recipient sites, the free edge of the folded greater omentum can simply be moved to the recipient site. If movement of the omentum to a distant recipient site is needed, the length of the omentum can be extended by “unfolding” the double layer after freeing the dorsal layer.1 The omentum and spleen are exteriorized and the dorsal omental layer is reflected ventrally and cranially to identify the pancreatic attachments (Figure 23-9). The dorsal layer is freed from the pancreas using sharp dissection proceeding from right to left. As the dissection approaches the tail of the left lobe of the pancreas, vessels entering the spleen are encountered. Several omental vessels originating from the splenic artery may be encountered and are ligated close to the spleen. Hemorrhage is controlled with ligation, radiosurgery or ligature clips. Care must be taken to avoid hematoma formation which will compromise the vascularity of the omentum.1 The omentum is now unfolded and extended caudally (Figure 23-10). The extension procedure provides considerable mobility and length with the free edge of the omentum reaching as far as the thoracic inlet cranially and the stifle caudally.1
Full extension of the omentum is achieved by making an inverted “L-shaped” incision through the dorsal extension and creating an omental pedicle (Figure 23-11). Beginning on the left side just caudal to the gastrosplenic ligament, the omental vessels are transected across one-half to two-thirds of the width. This makes the foot of the L incision. The incision should only be extended as far as necessary to preserve the width of the pedicle and subsequently the number of longitudinal omental vessels supplying the pedicle. The incision is then extended caudally and parallel to the remaining omental vessels. This incision forms the vertical limb of the L. The vertical incision can be extended as far as necessary, up to two-thirds of the length of the dorsal extension.
Hemorrhage is controlled with ligation, radiosurgery or ligature clips. The completed L-incision provides maximum extension of the omentum, reaching the muzzle cranially and the toes of the hindlimbs caudally.1 Microvascular transfer of a free omental graft is used frequently in people since extension of the omentum is restricted by the vascular pattern. Microvascular transfer of an omental graft to the distal extremity in dogs has been reported however ischemia and failure of the grafts occurred in three of the five dogs.5 Based on the success of omental extension described above, free transfer of the omentum is unlikely to be required in the dog or cat.
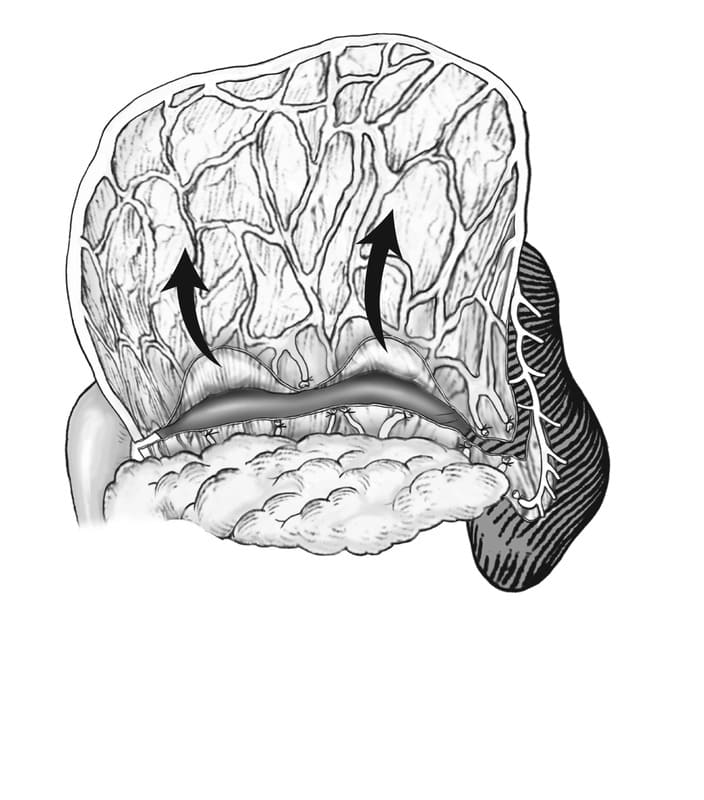
Figure 23-9. Dorsal extension of the omentum. The omentum and spleen are exteriorized, the dorsal leaf of the omentum is retracted ventrally and cranially and the omentum is freed from its pancreatic attachments. One or two vessels originating from the splenic artery are transected. (From Ross WE, Pardo AD. Evaluation of an omental pedicle extension technique in the dog. Vet Surg 1993;22:37-43.)

Figure 23-10. The dorsal leaf of the omentum is extended caudally. (From Ross WE, Pardo AD. Evaluation of an omental pedicle extension technique in the dog. Vet Surg 1993;22:37-43.)

Figure 23-11. Creation of the omental pedicle. The inverted L-shaped incision is begun just caudal to the gastrosplenic ligament (A) and extended caudally for full extension (B). (From Ross WE, Pardo AD. Evaluation of an omental pedicle extension technique in the dog. Vet Surg 1993;22:37-43.)
Extrabdominal Translocation of the Omentum
The omentum is most effectively translocated to the thoracic cavity by an incision in the diaphragm. Alternately, the omentum can be brought through the body wall, tunneled subcutaneously and then placed into the thoracic cavity through a lateral thoracic incision.
For other extrabdominal sites, the omentum is passed through an incision in the body wall, usually paracostal, and tunneled subcutaneously to the recipient site. Care must be taken to preserve the integrity of the omentum as it is grasped and tunneled through the subcutaneous tissue. Small tunnels that might constrict the omentum should be avoided.
Alternatively, the skin can be incised along the length of the tunnel and sutured over the length of the extrabdominal extension.
Specific Applications using the Omentum
Omentalization of Cystic Organs and Abscesses
The technique for using the omentum in the management of cysts and abscesses of abdominal organs is the same, regardless of the organ affected by disease. Extension of the omentum is usually not necessary for intraabdominal use. Omentalization of prostatic abscesses and cysts, pancreatic cysts and abscesses, uterine stump abscess, perinephric cysts, liver cysts and sublumbar lymph nodes has been reported.6-12 The technique requires the cyst to be partially or almost completely removed. The omentum is then packed into the remaining shell of the cyst and loosely sutured in place with monofilament, absorbable suture material. Abscess cavities are first cultured and drained and as much of the outer surface wall is removed as possible. Omentum is packed into the remaining abscess cavity and sutured in place.
Chylothorax
Translocation of the omentum into the thorax of a 6 year-old Rhodesian ridgeback15 and a 6-year-old Himalayan cat16 with chylothorax have been reported. The omentum was brought through an incision in the muscle of the diaphragm, spread out in the thorax and sutured in the region of the mediastinum. The basis for the use of the omentum in the treatment of chylothorax is to take advantage of the considerable lymphatic drainage provided by the omentum. Both animals were free of disease 16 and 13 months after surgery, respectively. Since the omental lymphatics drain into the cysterna chyli, the rationale for treatment of chylothorax with omental transposition has been questioned.17
In theory, non-chylous effusion that may occur following correction of chylothorax may be reduced following omentalization.
Chronic Wounds
Extraabdominal translocation of the omentum to sites of non-healing wounds has been reported in the dog and the cat.15-17 The omentum provides a vascular bed for free skin grafting or for random cutaneous or axial pattern skin flap reconstruction. The omentum is first extended and an omental pedicle is created if necessary. The omentum is then passed through a paracostal incision in the abdominal wall and tunneled subcutaneously to the wound bed. The skin is reconstructed over the omentum. Wound drainage with a closed, active drain may be required.
Thoracic and Abdominal Wall and Diaphragm Reconstruction
The omentum has been used in conjunction with mesh reconstruction of thoracic and abdominal wall defects.23,24 The omentum is first extended and an omental pedicle is created if necessary. The omentum is then passed through a paracostal incision in the abdominal wall and tunneled subcutaneously to the defect and placed on top of the mesh.
The omentum fills the soft tissue defect and also brings vascularity and lymphatic drainage to the wound site. The skin is reconstructed over the omentum. Wound drainage with a closed, active drain may be required.
The omentum has been used to cover experimentally created defects in the diaphragm in the dog. The omentum was folded over on itself and sutured to the edges of defect. After three weeks, the omentum remained viable with evidence of fibrometaplaisa with fibrosis to the diaphragm edges. Despite promising experimental results, reconstruction of the diaphragm is rarely required in dogs and cats. Primary closure of tears in the diaphragm is usually possible, even in chronic cases.
Other Applications
Application of the omentum to other surgical situations is possible, based on the ability of the omentum to enhance vascularity, lymphatic drainage and immune response at a recipient site. Use of the omentum to pack traumatic fractures in parenchymatous organs such as the liver, kidney and spleen is reported in humans. Support of gastrointestinal anastomotic sites is possible and widely used. Autgenous omental grafts, as free non-vascularized grafts, have been used to enhance bone healing in experimental non-union models in dogs.25,26 Radial osteotomies treated with free autogenous omentum had union by 16 weeks while untreated osteotomies remained as a non-union.25 In a second study using a similar non-union model in dogs, the effect of the omental graft was potentiated by the inclusion of adipose-derived stem cells.26 The surgical applications for use of the omentum are numerous and varied however, the use of omentum should be viewed as an adjunct to sound surgical practice and not as a substitute for good surgical technique.
References
- Ross WE, Pardo AD. Evaluation of an omental pedicle extension techinque in the dog. Vet Surg 1993;22:37-43.
- Evans HE. The abdomen. Millers Anatomy of the Dog. Philadelphia: W.B. Saunders Co; 1993:425.
- Gravenstein H. Uber die arterien des grossen netzes beim hunde. Morph Jahrb 1938;82:1-26.
- Nylander G, Tjernberg B. The lymphatics of the greater omentum: An experiemental study in the dog. Lymphology 1969;1:3-7.
- Roa DM, Bright RM, Daniel GB, McEntee MF, Sackman JE, Moyers TD. Microvascular transplantation of a free omental graft to the distal extremity in dogs. Vet Surg 1999;28:456-465.
- Bray JP, White RAS, Williams JM. Resection and omentalization: A new technique for management of prostatic retention cysts in dogs. Vet Surg 1997;26:202-209.
- Campbell BG. Omentalization of a nonresectabe uterine stump abscess in a dog. J Am Vet Med Assoc 2004;224:1799-1803.
- Friend EJ, Niles JD, Williams JM. Omentalisation of congenital liver cysts in a cat. Vet Rec 2001:149:275-276.
- Hill TP, Odesnik BJ. Omentalisation of perinephric pseudocysts in a cat. J Sm Anim Pract 2000;41:115.
- White RAS, Williams JM. Intracapsular prostatic omentalization: A new techinque for management of prostatic abscesses in dogs. Vet Surg 1995;24:390-395.
- Hoelzler MG, Bellah JR. Omentalization of cystic sublumbar lymph node metstases for long-term palliation of tenesmus and dysuria in adog with anal sac carcinoma. J Am Vet Med Assoc 2001;219:1729-1731.
- Johnson MD, Mann FA. Treatment for pancreatic abscesses via omentalization with abdominal closure versus open peritoneal drainage in dogs: 15 cases (1994-2004). J Am Vet Med Assoc 2006;228:397-402.
- Jerram RM, Warman CG, Davies ES, Robson MC, Walker AM. Successful treatment of a pancreatic pseudocyst by omentalisation in a dog. N Z Vet J. 2004;52:197-201.
- Franklin AD, Fearnside SM, Brain PH. Omentalisation of a caudal mediastinal abscess in a dog. Aust Vet J 2011;89:217-220.
- Williams JM, Niles JD. Use of omentum as a physiologic drain for treatment of chylothorax in a dog. Vet Surg 1999;28:61-65.
- LaFond E, Weirich WE, Salisbury SK. Omentalization of the thorax for treamment of idiopathic chylothorax with constrictive pleuritis in a cat. J Am An Hosp Assoc 2002;38:74-78.
- Sicard GK, Waller KR, McAnulty JF. The effect of cisterna chyli ablation combined with thoracic duct ligation on abdominal lymphatic drainage. Vet Surg 2005;34:64-70.
- Brockman DJ, Pardo AD, Conzemius MG, Cabell LM, Trout NJ. Omentum-enhanced reconstruction of chronic nonhealing wounds in cats: Techniques and clinical use. Vet Surg 1996;25:99-104.
- Lascelles BDX, White RAS. Combined omental pedicle grafts and thoracodorsal axial pattern flaps for the reconstruction of chronic, nonhealing axillary wounds in cats. Vet Surg 2001;30:380-385.
- Smith BA, Hosgood G, Hedland CS. Omental pedicle used to manage a large dorsal wound in a dog. J Sm Anim Pract 1995;36:267-270.
- Gray MJ. Chronic axillary wound repair in a cat with omentalisation and omocervical skin flap. J Small Anim Pract. 2005;46:499-503.
- Roa DM, Bright RM, Daniel GB, McEntee MF, Sackman JE, Moyers TD. Microvascular transplantation of a free omental graft to the distal extremity in dogs. J Small Anim Pract. 1998;39:475-480.
- Bright RM, Thacker LH. The formation of an omental pedicle flap and its experimental use in the repair of a diaphragmatic rent in the dog. J Am An Hosp Assoc 1982;18:283-289.
- Liptak JM, Dernell WS, Rizzo SA, Monteith GJ, Kamstock DA, Withrow SJ. Reconstruction of chest wall defects after rib tumor resection: a comparison of autogenous, prosthetic, and composite techniques in 44 dogs. Vet Surg 2008;37:479-487.
- Saifzadeh S, Pourreza B, Hobbenaghi R, Naghadeh BD, Kazemi S. Autogenous greater omentum, as a free nonvascularized graft, enhances bone healing: an experimental nonunion model. J Invest Surg. 2009;22:129-137.
- Bigham-Sadegh A, Mirshokraei P, Karimi I, Oryan A, Aparviz A, Shafiei-Sarvestani Z. Effects of adipose tissue stem cell concurrent with greater omentum on experimental long-bone healing in dog. Connect Tissue Res. 2012;53:334-342.
Get access to all handy features included in the IVIS website
- Get unlimited access to books, proceedings and journals.
- Get access to a global catalogue of meetings, on-site and online courses, webinars and educational videos.
- Bookmark your favorite articles in My Library for future reading.
- Save future meetings and courses in My Calendar and My e-Learning.
- Ask authors questions and read what others have to say.
About
How to reference this publication (Harvard system)?
Author(s)
Copyright Statement
© All text and images in this publication are copyright protected and cannot be reproduced or copied in any way.Related Content
Readers also viewed these publications
Buy this book
Buy this book
This book and many other titles are available from Teton Newmedia, your premier source for Veterinary Medicine books. To better serve you, the Teton NewMedia titles are now also available through CRC Press. Teton NewMedia is committed to providing alternative, interactive content including print, CD-ROM, web-based applications and eBooks.
Teton NewMedia
PO Box 4833
Jackson, WY 83001
307.734.0441
Email: [email protected]




Comments (0)
Ask the author
0 comments